

Hydrogen is an ideal fuel in many respects. It is the simplest atomic element, with a single proton and electron, the most plentiful element in the universe, and it has high energy content — three times more energy than gasoline on a per-pound basis. However, hydrogen is not a very practical fuel. It is not typically found alone on earth, but instead, is usually combined with other elements such as oxygen and carbon. Also, it is a gas at room temperature, thereby presenting handling, packaging and storage challenges.
UltraCell has pioneered RMFC technology for portable power applications. Methanol is a liquid at room temperature, and it is much easier to handle, package and store than hydrogen, making it a more practical fuel source. UltraCell uses its proprietary technology to reform the liquid methanol into hydrogen gas which is supplied directly to the fuel cell for electrical power production. Byproducts of the reforming process are small amounts of water vapor (H2O) and carbon dioxide (CO2), approximately equivalent to a child exhaling. The UltraCell reformed methanol fuel cell combines the practical advantages of methanol fuel packaging, storage and delivery with the energy advantages of hydrogen, and allows for a smaller and lighter weight power source for portable electronic devices.
Methanol is a hydrocarbon comprised of carbon, hydrogen and oxygen. Its chemical formula is CH3OH. Methanol is also known as wood alcohol because it was once produced chiefly as a byproduct of wood distillation. Today methanol is most often derived from the methane component of natural gas. It also can be produced from organic materials, and the resulting organic methanol, or bio-alcohol, is a renewable alternative to petroleum-based hydrocarbons. Methanol is biodegradable, dilutes quickly in large bodies of water, and is considered a clean alternative fuel by the U.S. Environmental Protection Agency.
For more information, go to: www.epa.gov
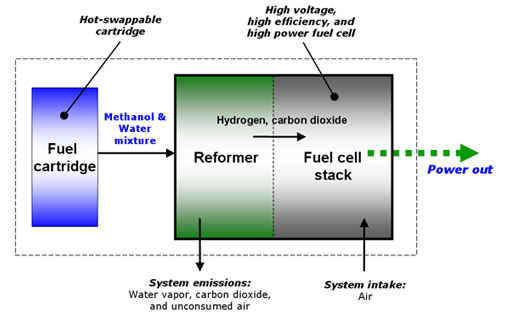 |
Overview of the Reformed Methanol Fuel Cell (RMFC) system |

